Groundwater Fluoride Exposure and Potential Effects on Arizona Livestock
Fluoride (F-) is a naturally occurring mineral commonly found in surface and groundwater. It is present in water sources due to the erosion of F- containing rocks and soil found throughout the United States. The natural presence of F- in groundwater, along with its proven dental health benefits, has led to its controlled addition to public water supplies where it is maintained at appropriate concentrations to help prevent tooth decay (Newbrun, 1989). While concentrations less than 2.0 parts per million (ppm) can be beneficial to both humans and animals, ingestion of F- over this concentration for many years may contribute to health challenges, specifically fluorosis (Blakley, 2021; World Health Organization, 2019). Fluorosis is a condition that affects bones and teeth in humans and various species of wild and domestic animals, including livestock. According to the U.S. Geological Survey (USGS) National Water-Quality Assessment (NAWQA) Program, F- was present at concentrations exceeding the Safe Drinking Water Act Secondary Maximum Contaminant Level (SMCL) threshold of 2.0 ppm in 12% of tested water sources in southwest U.S. states (Nevada, California, Arizona, Utah, and adjacent states) (Musgrove & Belitz, 2016). For reference, 1.0 ppm is roughly equivalent to a pinch of salt in one large bathtub (approximately 50 gallons) (Figure 1). Through the Safe Drinking Water Act, the U.S.
Environmental Protection Agency (USEPA) established the National Primary Drinking Water Regulations that set standards for specific chemicals that may cause human health impacts; this enforceable standard is the Maximum Contaminant Level. The USEPA also established National Secondary Drinking Water Standards that sets non-mandatory (that is, not enforceable) standards (known as secondary MCLs) for 15 chemicals to assist drinking water providers in managing the taste, color, and odor of their drinking water. While F- has a secondary MCL of 2.0 ppm, public water systems are required to publish notice to customers when F- concentrations in water exceed 2.0 ppm.
The USGS also observed that about 10% of Arizona wells exceeded the SMCLs (Musgrove & Belitz, 2016). Arizona ranks among the top five states where F- concentrations in public-supply and domestic wells exceed the SMCL and exceed Safe Drinking Water Act Maximum Contaminant Level (MCL) of 4.0 ppm (McMahon et al., 2020). As a result, a contemporary understanding of groundwater F- concentrations across Arizona is critical for human and livestock health. Here we focus on potential F- exposure for Arizona livestock.

Figure 1. Visual representation of 1.0 part per million (ppm).
Created in BioRender. Whitmore, C. (2025) https://BioRender.com/dagh9ak.
Jump To:
- Geography of fluoride in Arizona groundwater
- Importance of livestock in Arizona
- Routes of exposure for livestock
- Potential health risks
- Fuoride impacts to human health via meat consumption
- Acceptable fluoride ingestion for humans and livestock
- Removing fluoride from water
- Conclusions
- Acknowledgements
- Key terms
Geography of fluoride in Arizona groundwater
Previous publications by the Arizona Department of Environmental Quality reported the occurrence of F- in groundwater in southeast and southwest Arizona (Towne, 2006; 2011; 2015; Towne & Freak, 1999). Concentrations of F- in groundwater exceeded regulatory limits in older groundwater depleted of calcium, or in water from volcanic and sedimentary rock formations (Towne, 2011).
Fluoride concentrations in Arizona’s groundwater vary by hydrologic basin, reflecting the state’s diverse geology and hydrology. Our team extracted data from the USGS using the National Water Information System and mapped the available data (Figure 2). The results indicated that Fconcentrations exceeding 4.0 ppm (the Safe Drinking Water Act MCL) occur most frequently in groundwater found in southwestern Arizona and the southeastern border area with New Mexico. Additionally, the data further indicate that more than 10% of tested groundwater sources in basins in the western and southeastern areas of the state exceed the SMCL (2.0 ppm F-). This suggests potential for F- exposure via untreated groundwater for both livestock and people.

Figure 2. Map of fluoride occurrence by groundwater basin for the state of Arizona. Panel A represents the percentage of water samples exceeding 4.0 ppm (the Safe rinking Water Act Maximum Contaminant Level for people). Panel B represents the percentage of water samples exceeding 2.0 ppm (the recommended limit for fluoride in water for livestock). Data were obtained from U.S. Geological Survey National Water Information System.
Importance of livestock in Arizona
Livestock are a key sector of Arizona’s economy, especially in rural and Tribal communities. Cattle ranching is the most prominent type of livestock production in the state. According to the USDA National Agricultural Statistics Service Arizona Annual Bulletin, from 2015 to 2020, animal agriculture products contributed approximately $1.7 billion to Arizona’s economy. Beef and dairy cattle together accounted for about 46% of total meat animal sales and 46% of dairy product (milk) sales on average during that period (U.S. Department of Agriculture, National Agricultural Statistics Service, 2021). Arizona’s mild climate and wide-open rangelands make it ideal for year-round grazing (Sayre, 1999).
While Arizona ranchers have established smaller-scale production of sheep, goats, and poultry, cattle ranching remains dominant. Many Indigenous communities in the West have built strong livestock economies that have shaped their economies (Guice, 1977; Zappia, 2012). Cattle and other livestock have become integrated into Indigenous lifeways, helping to sustain food security and economic resilience (Lezama-Núñez et al., 2018). For many Indigenous communities, cattle ranching has long been tied to land stewardship, cultural identity, and generational knowledge. Water is essential to livestock operations, but it can be challenging to access, particularly when local groundwater quality does not meet recommended guidelines for livestock operations (described in greater detail below).
Routes of exposure for livestock
Cattle ingest F- through soil and feed, forage, and drinking water. Fluoride may enter cattle feed through phosphate-based mineral supplements derived from Frich rocks, from using water with high F- concentrations used to mix or moisten feed, or via crops and forage grown in soil containing F-. In the United States, there are drinking water standards to protect health. In contrast, the Canadian Council of Ministers of the Environment (CCME) has set a soil quality guideline at 400 ppm Ffor residential areas (Olkowski, 2009; Schuppli, 1985). Additionally, industrial pollution near grazing areas or feed sources may contribute to contamination. Industries such as aluminum and steel production may release Fcompounds into the atmosphere as either gases or particles (Choubisa, 2023). These pollutants may then settle onto nearby environments, contaminating soil, forage (such as grass and crops), and water sources used in grazing areas. As a result, cattle may ingest elevated F- by grazing on contaminated pastures or consuming feed and water from these affected sources. The chronic ingestion of water or feed with high F- concentrations highlights the importance of managing livestock exposure to protect both animal health and food safety (Choubisa, 2023; Whitford,1996).
Cattle ingest F- primarily through drinking water and feed, as illustrated in Figure 3. Once consumed, F- is absorbed in the gastrointestinal (digestive) tract and distributed via the bloodstream to various tissues, primarily accumulating in bones and teeth (Krook & Maylin, 1979). Once ingested and absorbed, it will accumulate in bones and teeth due to its high affinity for calcium, which leadsto its incorporation into the hydroxyapatite structure of bones and teeth (Whitford, 1996). Namely, hydroxide ions are replaced with F- ions (Figure 4), which can strengthen the enamel, but with excessive F- exposure, may lead to brittle teeth. Fluoride accumulation in soft tissues is generally low because of its preference for bone and teeth, (Shupe et al., 1992; Suttie et al., 1972). Over time, the F- bioaccumulation can negatively affect cattle health, causing mobility issues and reduced productivity, which can ultimately result in economic losses for ranchers.

Figure 3: Examples of ingestion exposure route (with possible sources) and sites of accumulation in the gastrointestinal (digestive) tract and teeth of cattle.
Created in BioRender. Whitmore, C. (2025) https://BioRender.com/rif2t7z.
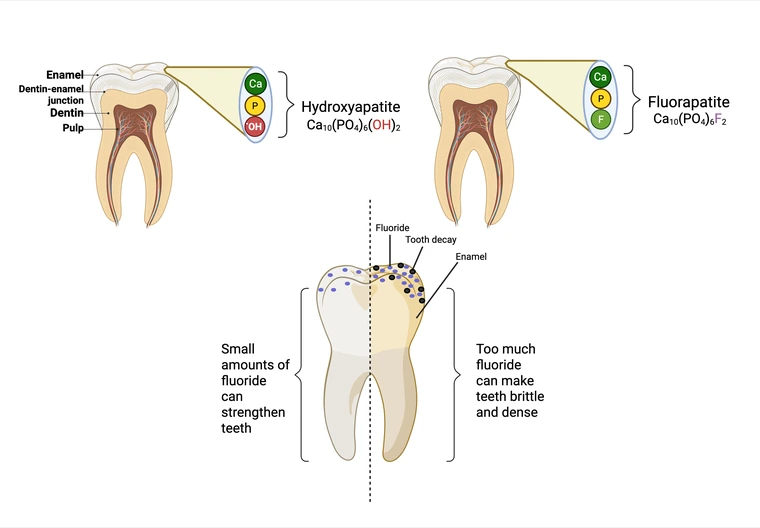
Figure 4. Mechanism of fluoride incorporation into tooth enamel. Hydroxyapatite is a mineral form of calcium apatite that comprises most of the bone structure in teeth and bones. Fluorapatite is formed when hydroxide ions (OH-) are replaced with F- ions. Excessive F- can cause damage to the tooth enamel making the teeth more porous and susceptible to tooth decay.
Created in BioRender. Whitmore, C. (2025) https://BioRender.com/zzznwrf
Potential health risks
Fluorosis is a form of chronic F- toxicity that is caused from long-term exposure to excessive F- concentrations. Fluoride toxicity occurs in either acute or chronic forms; however, chronic cases in cattle is uncommon in the United States under normal management practices (Blakley, 2021; Suttie, 1977). However, it may occur in specific situations, motivating the need for knowledge about F- concentrations in water and feed to reduce animal exposure. Fluoride exposure may begin early in life as calves ingest water and feed containing F- (Krook & Maylin, 1979). Cattle are susceptible to F- accumulation during the rapid growth phases, particularly from weaning to maturity. Fluoride can cross the placental barrier, and some of it is stored in the developing calf's bones (Greenwood et al., 1964). The amount stored in the calf’s bones are linked to how much F- is consumed by the dam, the calf’s mother, and the amount in her blood during pregnancy (Greenwood et al., 1964). Cattle may show signs of F- toxicity in as little as 6-12 months, especially when exposure begins as calves. However, more significant signs of fluorosis will often develop when cattle are one to three years of age, which may align with the typical slaughter ages for beef cattle. In conventional grain-finished systems, beef cattle are typically slaughtered between 18 and 24 months of age. In grass-finished systems, cattle can be slaughtered up to 30 months of age (Garcia et al., 2008). Chronic F- exposure may lead to gradual accumulation in bone, potentially causing dental and skeletal fluorosis. Dental fluorosis, characterized by discoloration and erosion of teeth, is often the first visible sign of chronic F- exposure. This condition can impair grazing efficiency and feed intake, indirectly affecting growth and productivity (Whitford, 1996). Skeletal fluorosis results from prolonged exposure to high F- concentrations, leading to bone deposition. Symptoms include joint stiffness, lameness, and bone fractures, severely impacting the cattle's mobility and overall wellbeing (Suttie & Phillips, 1960).
Fuoride impacts to human health via meat consumption
To date, no studies have identified any human health risk associated with consuming meat tissue from cattle that ingest high concentrations of F- through their water, feed, or crops. In the U.S., studies have predominantly focused on F- exposure through water rather than directly assessing its accumulation in cattle meat or other dietary sources. Additionally, F- can accumulate in crops grown in contaminated soil, further contributing to cattle exposure. A critical information gap remains on conducting risk assessments that combine F- from well water, crops, cattle exposure, and human consumption.
Acceptable fluoride ingestion for humans and livestock
There are no federally enforced F- standards for livestock feed in the United States; however, guidelines may be derived from scientific literature. Research suggests that the maximum recommended concentration of F- in feed for beef cattle is approximately 50 ppm (Greenwood et al., 1964; Synek et al., 1978), with higher limits generally tolerated by dairy cattle and lower limits recommended for sheep (Table 1). Sheep may be particularly sensitive to F-; one study reported that dietary concentrations as low as 30 ppm can disrupt normal bone remodeling and calcium replacement in the bone matrix, resulting in structural abnormalities such as brittle bones (Said et al., 1977). In most food-producing animals, excluding sheep, chronic F- poisoning is typically associated with dietary concentrations ranging from 100 to 300 ppm, depending on the duration and conditions of exposure.
While there is no comprehensive federal livestock water standard for F-, research literature provides evidence to support general guidelines. Furthermore, individual states may adopt their own F- guidelines for air, water, food and other media. For example, the state of Kansas has established a F- guideline of 2.0 ppm in livestock water (Agency for Toxic Substances and Disease Registry, 2003). Prolonged consumption of water with F- concentrations exceeding 4.0 ppm may pose health risks to livestock, particularly long-lived breeding animals. The drinking water standard promulgated by the USEPA through the Safe Drinking Water Act to protect human health are presented in Table 2 alongside recommendations from the National Research Council and Olkowski (2009) for livestock water concentrations of F-.
The F- drinking water guideline for livestock (2.0 ppm) is lower than the human MCL (4.0 ppm) because animals, particularly grazing livestock, are exposed to F- from additional sources such as their feed or forage. Over time, this cumulative exposure increases the risk of dental or skeletal fluorosis. Therefore, a more protective limit is set for livestock to account for their higher total intake and to prevent adverse health effects such as dental and skeletal fluorosis.
Managing F- exposure in livestock is strengthened by testing livestock water and feed for F- to ensure cumulative intake is within acceptable limits. The state of Arizona maintains a list of certified laboratories for measuring F- and other contaminants in drinking water. The US Department of Agriculture Agricultural Marketing Services National Science Laboratories maintains a list of fee-for-service labs that will test feed and other materials for F-.
| Livestock | Fluoride in feed guidelines (ppm) |
|---|---|
| Beef cattle | 50.0 |
| Dairy cattle | 60.0-100.0 |
| Sheep | 10.0 |
| Finishing pigs/ Feeder lambs | 150.0 |
| Horses | 60.0 |
| Chickens | 300.0-400.0 |
| Organism | Fluoride in drinking water guidelines (ppm) |
|---|---|
| Human | 4.0a |
| Livestock (general) | 1.0-2.0b |
| Slaughter cattle | 12-15b,c |
| Mature beef cattle | 4-8b,c |
| Young dairy cattle | 2.5-4.0b,c |
| Mature dairy cattle | 3-6b,c |
Removing fluoride from water
The USDA does not currently recommend a specific method for F- removal from livestock wells. While most treatment methods in Table 3 are designed for household use, they may be adapted for livestock wells.
Reverse osmosis (RO)
A membrane filtration process commonly used for F- removal from water. This removes several contaminants, including F- ions, heavy metals, salts, and organic compounds, allowing only water molecules to pass through. The process is highly effective at reducing F- concentrations, with removal rates of up to 90-95%.
Ion exchange
A chemical process where ions, such as F-, are exchanged with other similarly charged ions, typically chloride, on the surface of resin beads. As water flows through the resin, F- ions are electrostatically chemically exchanged with chloride ions, effectively removing them from the water. While ion exchange is effective for F- removal, it is costly to replace resins.
Absorptive technologies
These include activated carbon, alumina, and other adsorbents. This process can be cost-effective and does not produce any concerning by-products. This method may be an effective option for both community and rural systems where maintenance can occur regularly.
More information on specific types of water treatment and questions to ask when purchasing treatment equipment can be found in Arizona Know Your Water (Artiola et al., 2012). If these treatment options are not practical or feasible, then managing exposure becomes critical: limit the amount of water the herd consumes from the affected well and rotate to alternative water sources or wells in the area.
| Removal method | Mechanism | Cost | Maintenance | Removal effectiveness |
|---|---|---|---|---|
| Reverse osmosis | Uses a semi-permeable membrane to filter out F and other ions | $$$ – High initial cost and maintenance costs ~$300–$800 for home units | Moderate to High – due to membrane maintenance; membrane needs cleaning or replacement every 1–2 years and filters every 6–12 months | Very High (90-95% removal) |
| Ion exchange | Fluoride ions are exchanged with hydroxide or chloride ions on resin surface | $$ – Medium cost ~$100–$500 depending on scale | Moderate – Requires occasional resin regeneration with chemicals | Moderate (60–90% removal) |
| Absorption | Fluoride binds to media like activated carbon without producing harmful byproducts | $ – Low initial cost ~$50–$200 for household systems | Moderate – Due to media replacement; replacement is needed every few months (depends on frequency of use and water quality) | Moderate to High (70–95% removal) |
Conclusions
While chronic, long-term F- exposure in livestock, particularly cattle, may lead to dental and skeletal fluorosis, the risk to human health through meat consumption remains low, though uncertain due to limited previous research. Despite this limitation, previous work has demonstrated that chronic F- accumulation in cattle, primarily through water, feed, forage, and crops, is an important consideration for livestock health. Due to its widespread occurrence in groundwater through Arizona, continued research is needed to address knowledge gaps regarding F- accumulation in cattle and refine our knowledge of how to effectively diminish risks to maintain livestock health and well-being.
Acknowledgements
Funding that supported this work was provided by the National Institutes of Health (P50MD015706, 2P42ES004940, P30 ES006694), US Geological Survey (G24AC00083) and the Waverly Street Foundation. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health, US Geological Survey, or Waverly Street Foundation.
Key terms
- Fluoride (F-): a naturally occurring element that is found in rock, soil, water, and other environmental media that can accumulate in plant.
- Fluorosis: a health condition that affects bones and teeth when exposed to excessive fluoride over a long period of time. ▪ Public water system – a system with 15 or more service connections or a system that services 25 or more people.
- Safe Drinking Water Act: a federal law passed by the US Congress in 1974 (42 U.S.C. §300f et seq.) to protect the quality of drinking water in the United States. The Act authorizes the US Environmental Protection Agency to promulgate minimum drinking water standards, enforce those standards, and compels public water systems to comply with USEPA set regulations.
- Maximum Contaminant Level (MCL): The highest level of a contaminant that is allowed in drinking water. The MCL is set by the USEPA through the National Primary Drinking Water Regulations and these standards are legally enforceable for all public water systems.
- Secondary Maximum Contaminant Level (SMCL): a non-enforceable standard set by the US EPA through the National Secondary Drinking Water Standards for 15 chemicals to assist drinking water providers in managing the taste, color, and odor of drinking water.
- Concentration: the amount of a dissolved substance that exists in a solution or mixture.
- Accumulation: the gradual buildup of an element over time.
- Route of exposure: the manner in which a contaminant or other material enters an organism’s body. The primary routes of exposure include inhalation (breathing), ingestion (eating or drinking), and dermal contact (skin contact).

